|
11/22/2023 0 Comments Timi trials 
Any condition that, in the opinion of the investigator, would make trial participation not in the best interest of the subject, or would compromise compliance with the trial protocol (e.g.For Marc Sabatine, MD, MPH, of the Division of Cardiovascular Medicine, going through all the clinical trials spearheaded by one of the Brigham’s premier cardiovascular research centers is like “keeping track of all of one’s children.” Since its founding by the U.S.Current participation in a clinical trial with an unlicensed drug or device.Women of child-bearing potential (unless using adequate contraception) or currently breastfeeding.History of heart failure due to restrictive or infiltrative cardiomyopathy, active myocarditis, constrictive pericarditis, hypertrophic (obstructive) cardiomyopathy, uncorrected primary valvular disease, complex congenital heart disease, or heart failure felt to be due to a transient process (e.g., stress cardiomyopathy, tachycardia-induced cardiomyopathy) expected to resolve within 2 months.History of heart transplantation or current transplant listing mechanical circulatory support use (either durable or temporary) during the index hospitalization.Untreated sustained ventricular arrhythmias or Mobitz type II or third-degree heart block (i.e., without an ICD or pacemaker, respectively).ST-segment elevation myocardial infarction or coronary revascularization (percutaneous coronary intervention or coronary artery bypass grafting) within 30 days prior to randomization or intent to undergo coronary revascularization during the trial.Implantation of a cardiac resynchronization therapy (CRT) device or valve repair or replacement within 30 days prior to randomization or intent to do so during the trial.(Only applies to patients with T2DM who are on insulin and/or a sulfonylurea) History of recurrent major hypoglycemia (i.e., resulting in severe impairment in consciousness or behavior, or requiring emergency external assistance).Type 1 diabetes mellitus or history of diabetic ketoacidosis.eGFR Concurrent use of two or more intravenous inotropic agents during the index hospitalization.Symptomatic hypotension in the past 24 hours.Trial leadership will monitor the proportion of patients with and without type 2 diabetes and may cap enrollment of one subgroup to ensure adequate representation of the other. In addition, patients with and without type 2 diabetes are eligible for participation in the trial. Trial leadership will monitor the proportion of patients with various LVEFs and may cap enrollment of certain subgroups to ensure a broad population. Patients across the spectrum of LVEF are eligible for participation in the trial. 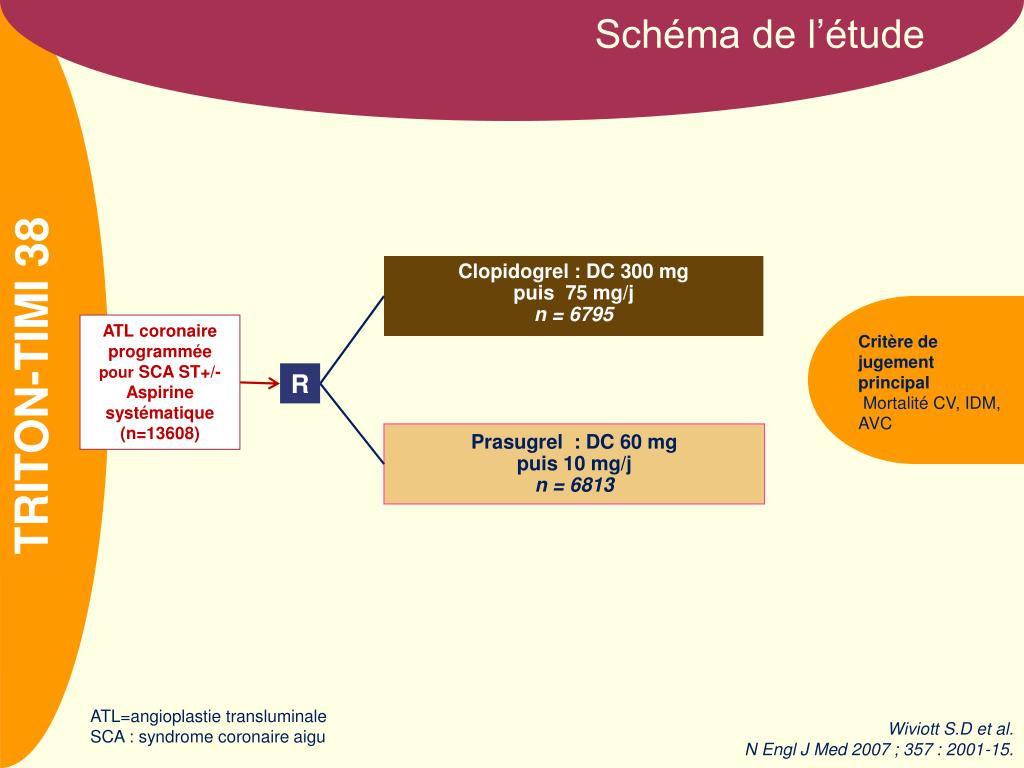
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
